Abstract
Introduction: Five-year overall survival after childhood lymphoma has steadily increased; however, patients continue to experience chemotherapy-related toxicities (chemotoxicities). We have previously demonstrated that lower skeletal muscle density (SMD) at diagnosis is associated with greater odds of ≥grade 4 hematologic toxicity (Wadhwa, Cancer 2021) in children with lymphoma and rhabdomyosarcoma. Body composition is likely to change after cancer diagnosis; however, the longitudinal trajectory of body composition after cancer diagnosis and the association of the change in body composition with chemotoxicity remains unknown. We examined these gaps in knowledge in a cohort of 87 children with lymphoma (Hodgkin lymphoma [HL]: n=45 and non-Hodgkin lymphoma [NHL]: n=42) treated at a single center between 2000 and 2015.
Methods: We used two consecutive computed tomography [CT] scans (first scan at diagnosis) to measure body composition (automated module [ABACS] of sliceOmatic [Tomovision, Quebec, Canada]). Using a single cross-sectional slice at the third lumbar vertebra, we calculated skeletal muscle index (SMI in cm2/m2), SMD (in Hounsfield Units) and height-adjusted total adipose tissue (hTAT, in cm2/m2) at both time points. Body-mass index (BMI, converted into percentile using CDC growth charts, BMI%ile) and body surface area (BSA) were also calculated at each time point. Demographics and clinical details including chemotoxicities (hematologic and non-hematologic, graded using CTCAE v5) during the first six months post-diagnosis were abstracted from electronic medical records. Changes in body composition from baseline to follow up CT scan were analyzed using generalized linear regression models adjusting for time between scans, age at cancer diagnosis, sex, race/ethnicity, primary cancer diagnosis and disease-specific risk stratification. Association between changes in body composition and chemotoxicity was evaluated using linear regression models.
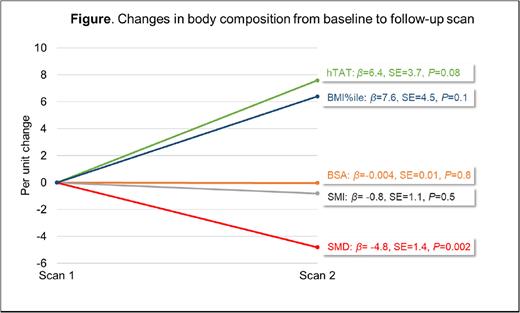
Results: Median age at diagnosis for the cohort was 12.9y (range 2-18.6). The majority of patients were non-Hispanic white (59.8%) and male (60.9%). Of the 87 patients with available baseline CT scan (scan 1), 62 (71%) had a follow-up scan (scan 2); the median interval between the two scans was 45 days (range, 8-207). Longitudinal change in body composition: Figure highlights changes in body composition (BMI%ile, BSA, SMI, SMD and hTAT) after adjusting for covariates. SMD showed a significant decline from scan 1 to scan 2 (βscan1_scan2= -4.8, SE=1.4, P=0.002). hTAT showed a non-significant increase (βscan1_scan2=6.4, SE=3.7, P=0.08), as did BMI%ile (βscan1_scan2=7.6, SE=4.5, P=0.1). BSA and SMI did not show a change between scans 1 and 2. Prevalence and risk of chemotoxicity: With a median of 158 days from diagnosis (range, 66-223), we observed ≥grade 4 hematologic toxicities in 60% of the cycles and ≥grade 3 non-hematologic toxicities in 23.6% of the cycles. Per unit decline in SMD between scan 1 and scan 2 was associated with an increase in risk of ≥grade 3 non-hematologic toxicity (β=1.37; 95%CI=0.2-2.4, P=0.02) after adjusting for sex and race/ethnicity. We did not identify an association between change in SMD and hematologic toxicities (β=0.2; 95%CI=-0.9-1.3, P=0.7). We did not identify any association between other body composition metrics and chemotoxicity.
Conclusions: Children with lymphoma undergo a significant decline in SMD during early treatment, which may be associated with increased risk of chemotoxicities. Importantly, BSA (and hence chemotherapy dosing) remains stable, suggesting that other body composition metrics may need to be incorporated into chemotherapy dosing to optimize survival while reducing the risk of toxicity. Further validation of these findings in larger cohorts of uniformly treated patients with lymphoma across clinical trials is currently underway.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal